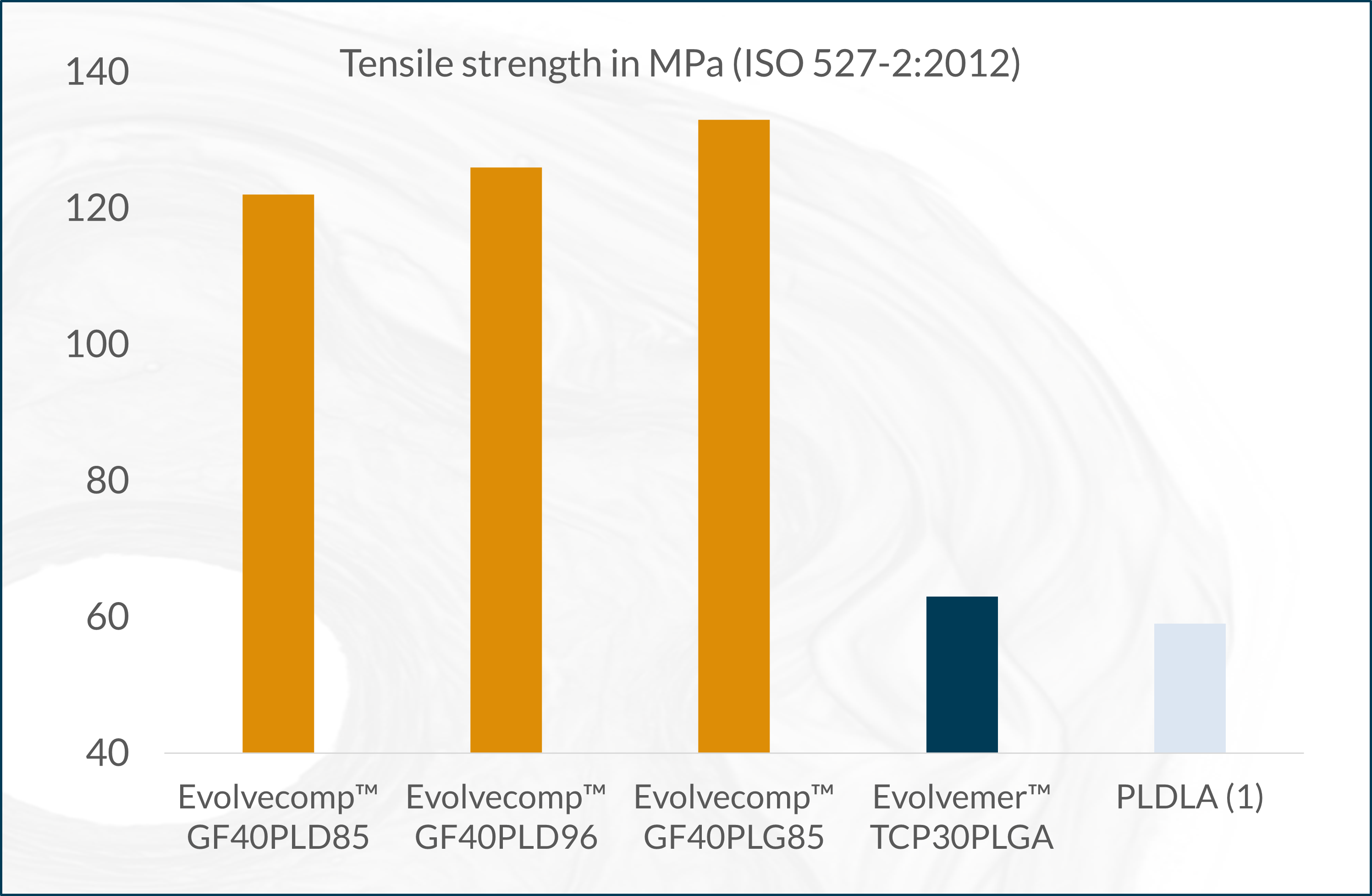
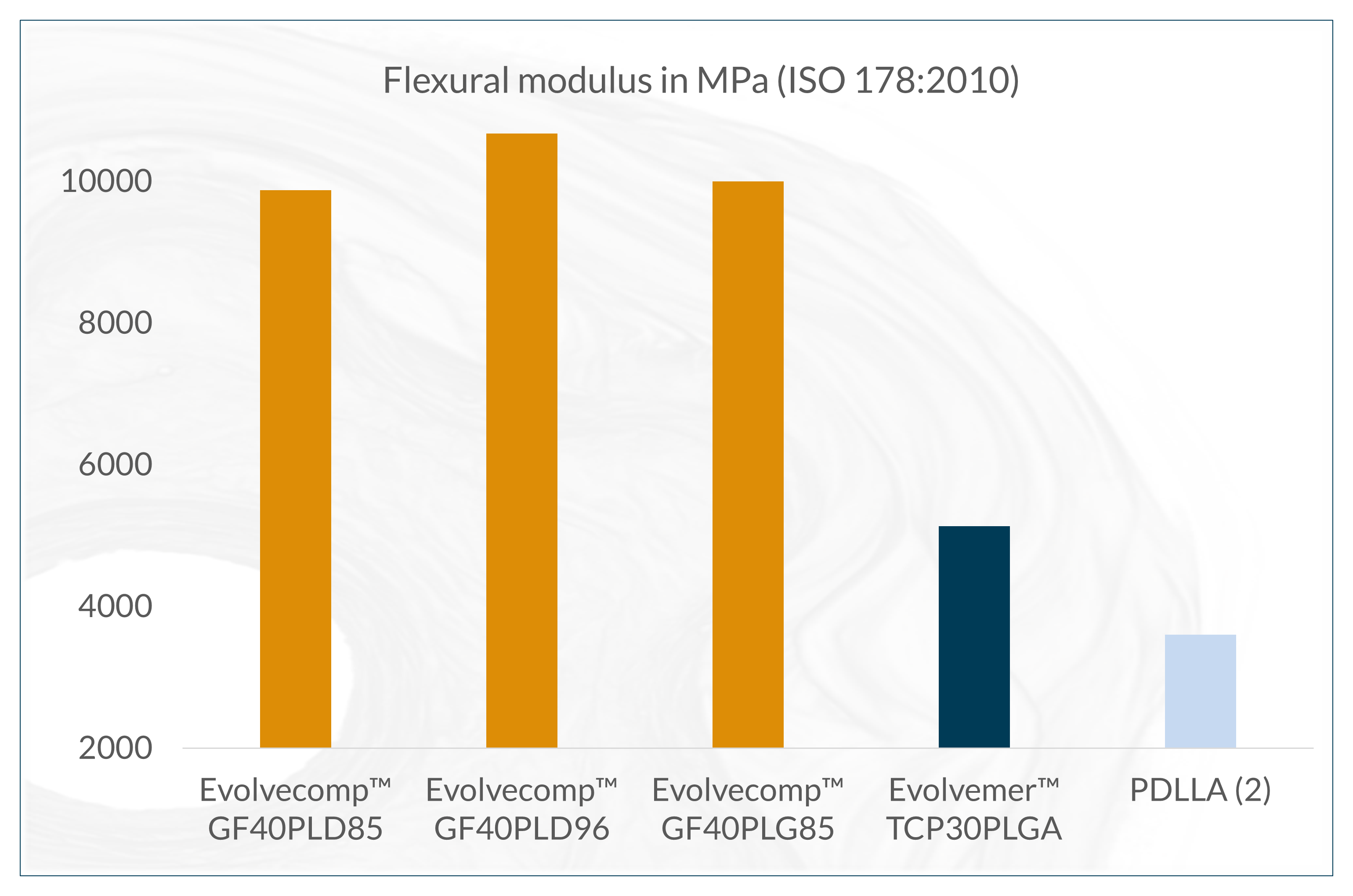
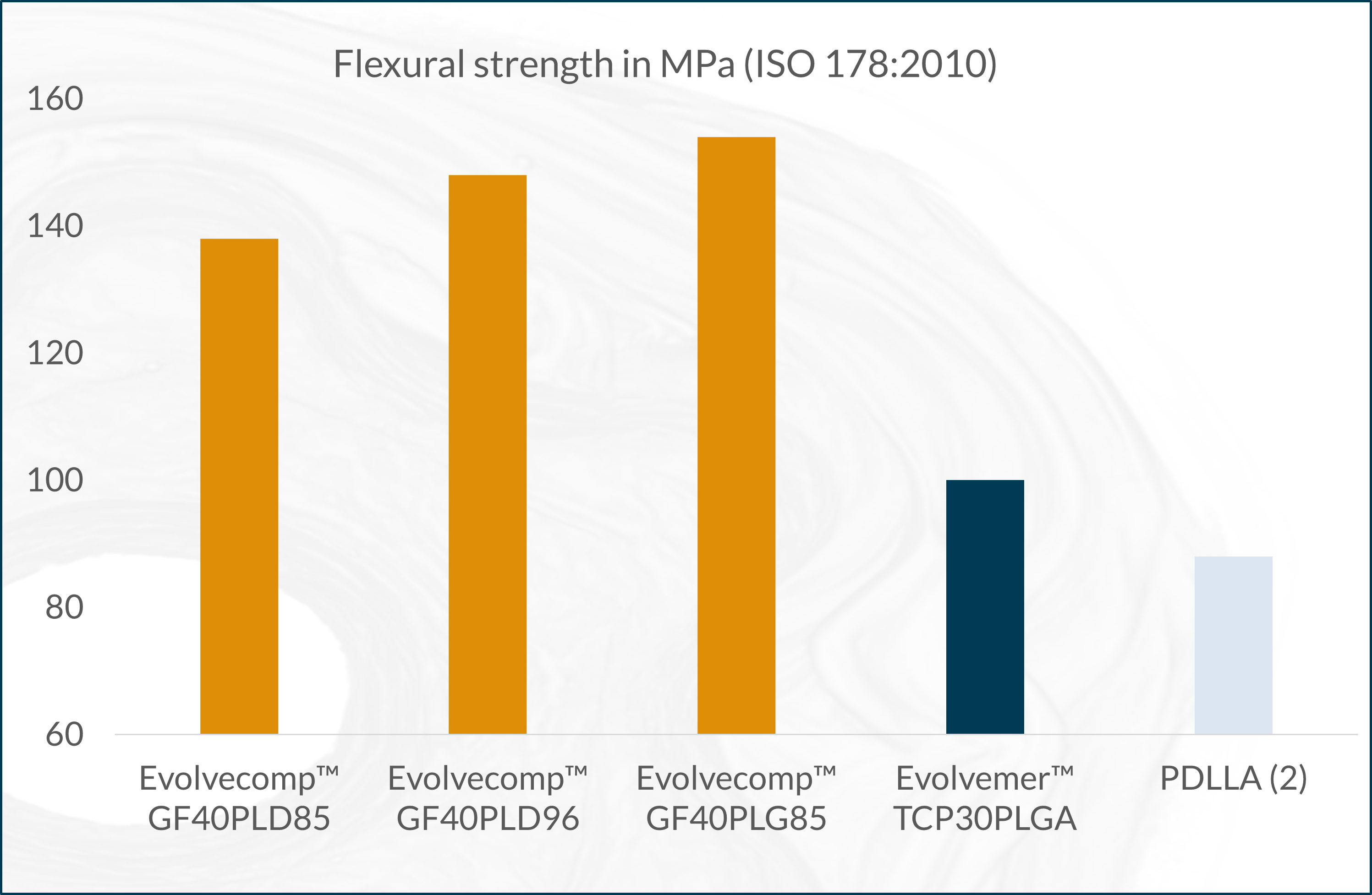
The next-generation bioabsorbable Evolvecomp™ composites are reinforced with bioactive natural mineral fibers, ABM´s X3 fibers. With these composites, our customers can realize their visions for strong bioabsorbable orthopedic implants. ABM´s unique bioactive natural mineral fiber (fibre), X3 provides an extremely strong and osteoconductive ingredient to combine with bioresorbable materials. Together the composite forms a raw material with excellent benefits even for load-bearing applications.
The X3 bioactive natural mineral fibers are osteoconductive, which allows direct bone-implant interaction and stimulation of osteogenesis. The absorption time can be tailored by composition selection and processing. The Evolvecomp™ grades are suitable for demanding injection moulding, extrusion and compression moulding applications.
All the medical grade Evolvecomp™ materials show excellent biocompatibility according to ISO 10993-1 standard. A balanced pH environment promotes bone healing and improves biocompatibility. More information about the biocompatibility.
Access the science behind the X3 fiber composite and read more at the White Papers and Studies section.
Technical Information
Evolvecomp GF40PLG85Evolvecomp GF40PLD85Evolvecomp GF40PLD96
Other compositions are also available on request.



(1) Salmoria G. V. et al. Properties of injection-molded poly (L-co-D, L-lactic acid) using different melt temperatures and stress concentrator in the specimen geometry. The International Journal of Advanced Manufacturing Technology (2018).
(2) Perego G. et al. Effect of molecular weight and crystallinity on poly(lactic acid) mechanical properties. Journal of Applied Polymer Science 59, 37-43, 1996.