Bioabsorbable Evolvemer™ TCP30PLGA is a biocompatible composite of bioabsorbable 85L/15G PLGA base polymer and beta-tri-calcium phosphate (β-TCP).
The β-TCP particle size distribution is optimized in this high-quality composite. This makes its processing easier and improves the mechanical properties of the final products.
Biocompatibility of the Evolvemer™ Composite has been evaluated according to ISO 10993-1:2009 (“Biological evaluation of medical devices – Part 1: Evaluation and testing within a risk management process”). Based on the evaluation, Evolvemer™ Composite presents no risk of systemic toxicological effects.
Cytotoxicity and Plastic Class V tests have been conducted for Evolvemer™ Composite to support the biocompatibility statement of the composite. However, it is the sole responsibility of the manufacturer of the final end-use product to determine the biocompatibility of the final product.
Technical Information
Evolvemer™ TCP30PLGA


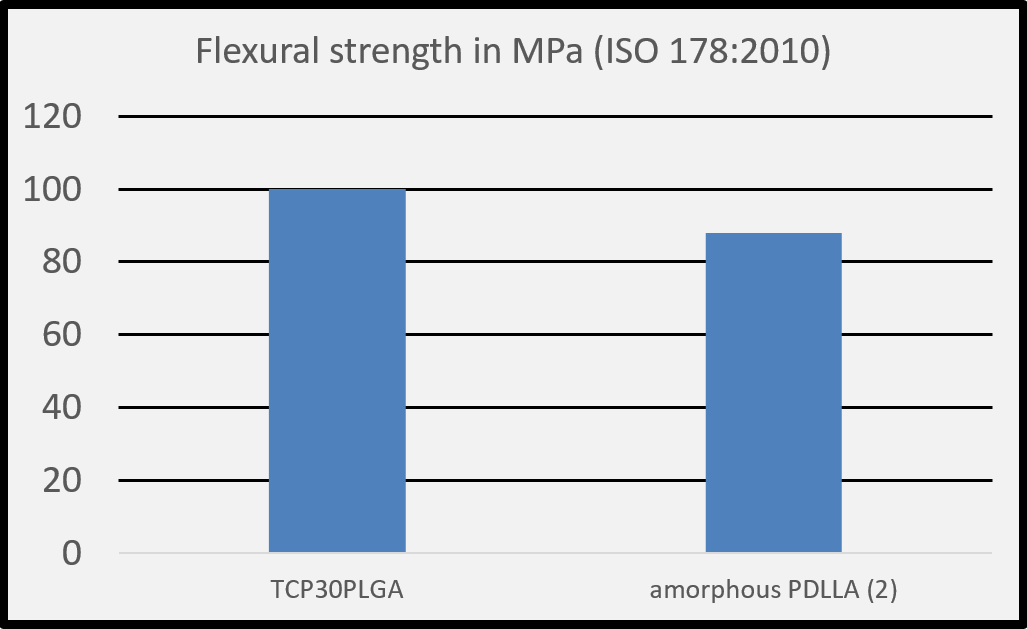
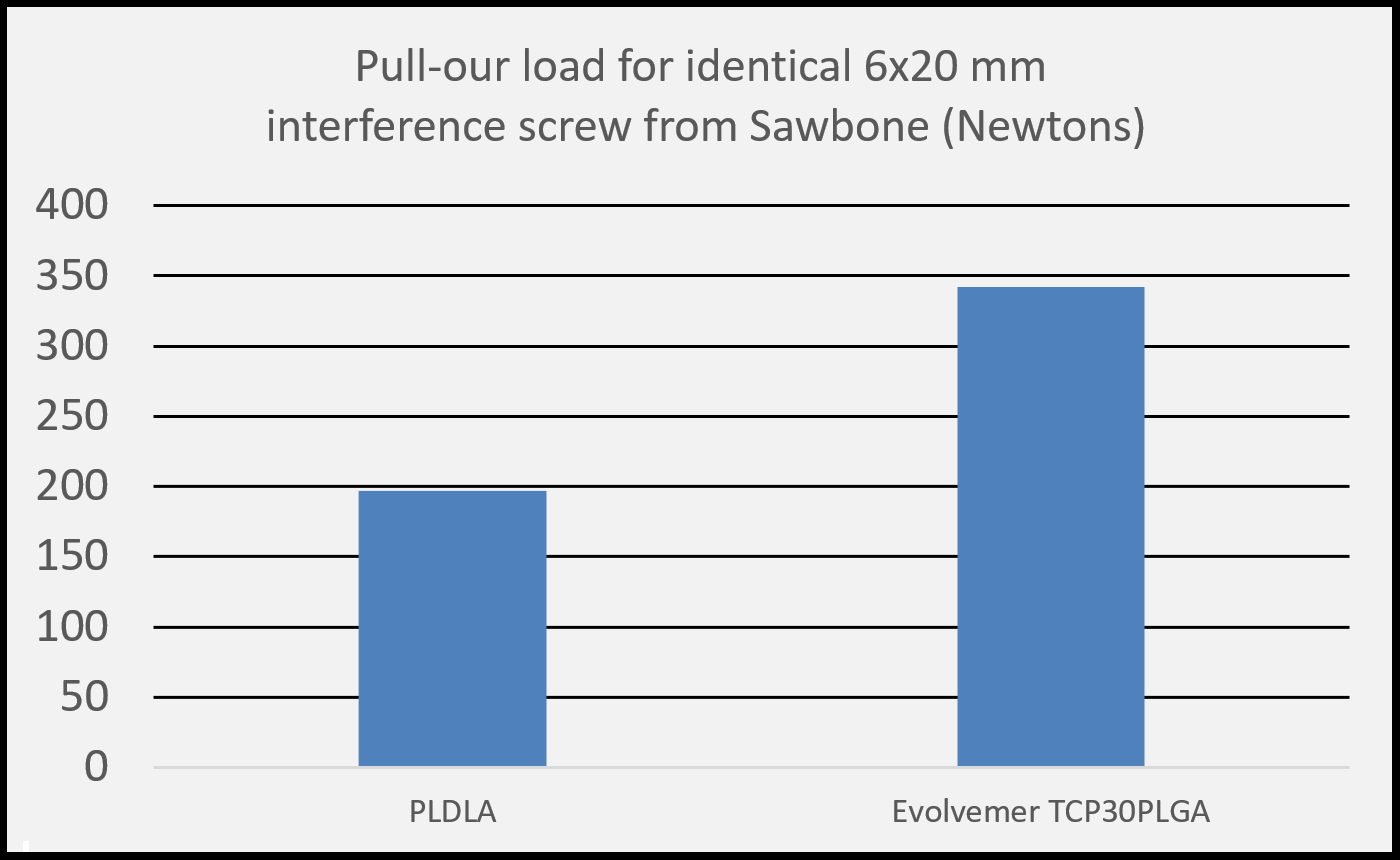
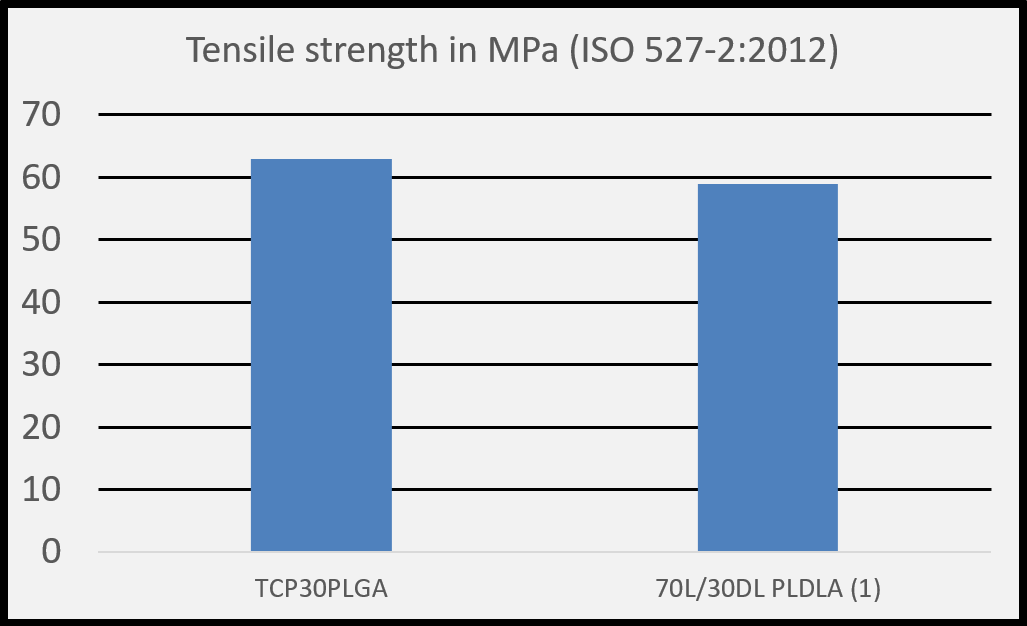
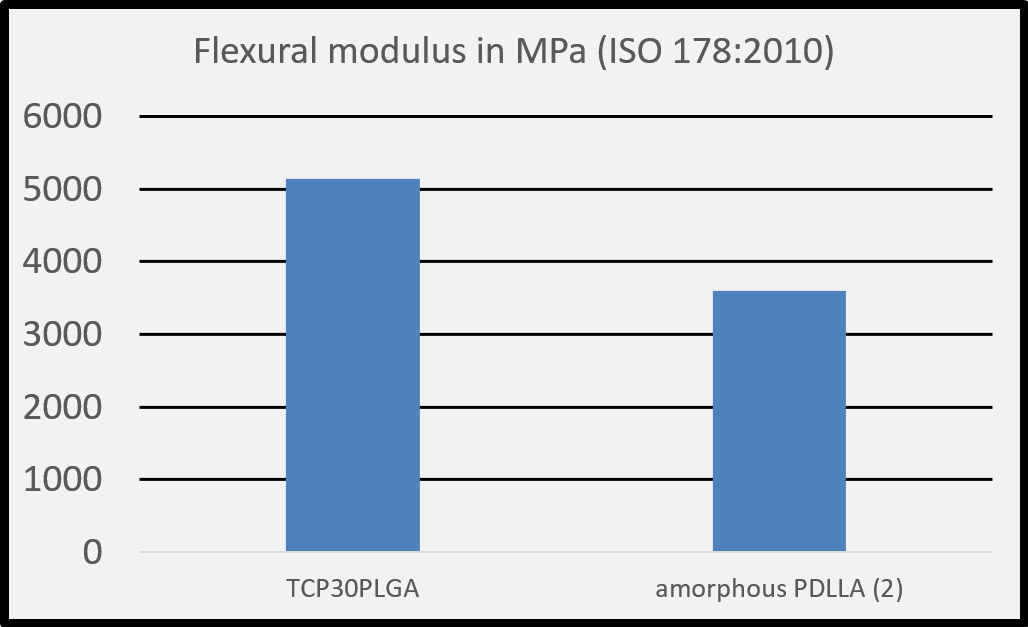
(1) Salmoria G. V. et al. Properties of injection-molded poly (L-co-D, L-lactic acid) using different melt temperatures and stress concentrator in the specimen geometry. The International Journal of Advanced Manufacturing Technology (2018).
(2) Perego G. et al. Effect of molecular weight and crystallinity on poly(lactic acid) mechanical properties. Journal of Applied Polymer Science 59, 37-43, 1996.




